|
EIN for organizations is sometimes also referred to as taxpayer identification number or TIN or simply IRS Number. The employer identification number (EIN) for 4d Molecular Therapeutics Inc is 473506994. The result is a customized, novel, and proprietary pharmaceutical-grade product candidate designed for targeted therapeutic gene delivery and efficacy in humans.4d Molecular Therapeutics Inc is a corporation in Emeryville, California. After defining the target product profile, and the associated target vector profile, 4DMT then applies proprietary methods to identify lead vectors from within our AAV libraries. 4DMT’s Therapeutic Vector Evolution platform can deploy over 35 unique and proprietary AAV libraries comprised of an estimated 1 billion vector capsid sequences. Peter Francis, Chief Medical Officer at 4DMT.Ī copy of the poster presentation can be found in the “Investors & News” section of the company website, under “Events & Presentations”: About 4DMT’s Therapeutic Vector EvolutionĤDMT is advancing the field of targeted and optimized AAV vector technology by deploying principles of evolution and natural selection to create vectors that are designed to efficiently and selectively target the desired cells within the diseased human organ via clinically optimal routes of administration, at manageable doses and with resistance to pre-existing antibodies in the population. 
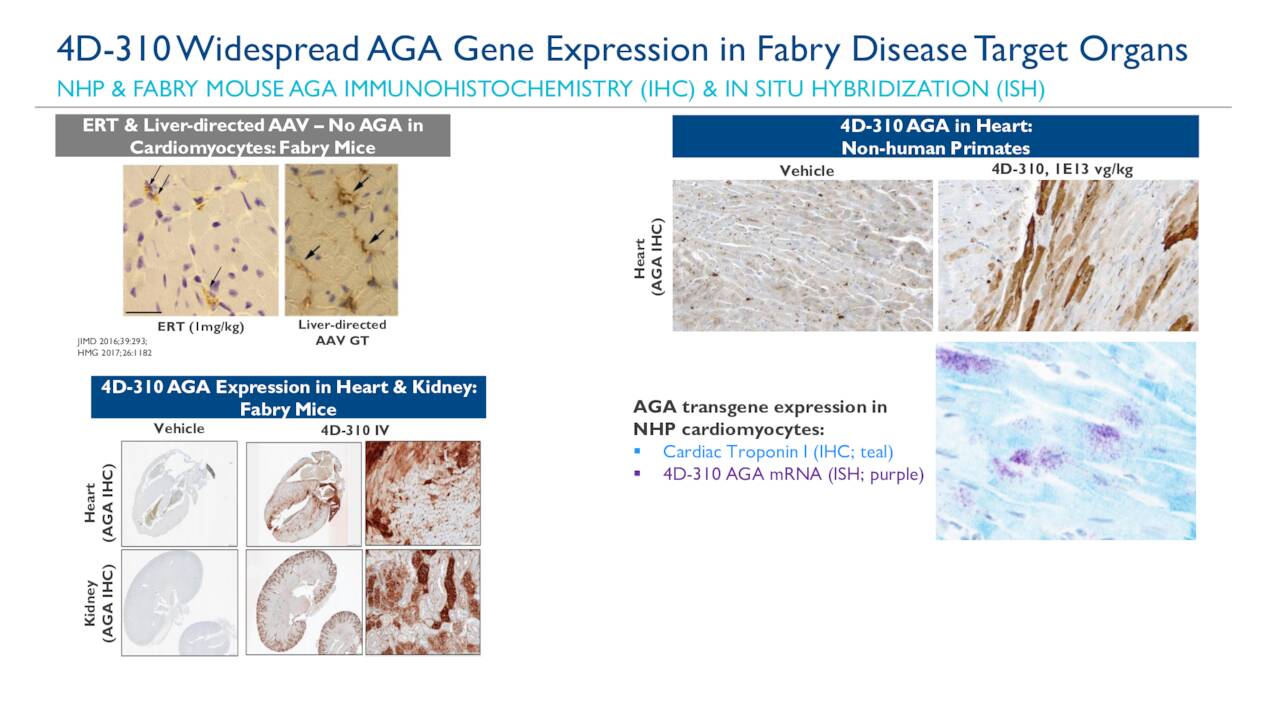
We are pleased to present these data as an early demonstration of our platform’s ability to potentially address neuromuscular diseases,” said Dr. “4DMT is building upon our Therapeutic Vector Evolution platform with the goal of ultimately bringing gene therapy products to patients with severe genetic diseases. Gabriel Brooks, VP Clinical Research and Development, and Program Leader, Neuromuscular and LSD Therapeutic Areas at 4DMT. This data package also provides the basis of a preclinical data package that we believe will enable commencement of clinical development of our Fabry product candidate,” said Dr. “The data generated using the novel vector, 4D-C102, and the 4D-310 Fabry product candidate using that vector, helps validate 4DMT’s Therapeutic Vector Evolution approach for in vivo cardiac tissue targeting. 4DMT’s Fabry product candidate, 4D-310, aims to preferentially express the GLA enzyme in cardiomyocytes, in addition to other tissues including kidney and liver, to directly address the cause of Fabry-related heart disease. As a result, heart disease is the most common cause of mortality in these patients. Current enzyme replacement therapy partially clears accumulated lipids from the endothelial cells of affected organs however, clearance in other cell types, including cardiomyocytes, appears incomplete. The data was highlighted in a poster presentation at the 6 th International Update on Fabry Disease held May 26 to 28, 2019 in Prague, Czech Republic.įabry disease is a rare, X-linked, monogenic disorder caused by mutations in the GLA gene that results in storage and accumulation of lipids and leads to debilitating effects on a wide range of organs and systems, including the heart. (4DMT), a leader in the discovery and development of targeted, customized and proprietary next-generation adeno-associated virus (AAV) vectors and gene therapy products, announced new preclinical data with its novel vector, 4D-C102, broadly for neuromuscular diseases and specifically for the product candidate incorporating 4D-C102 for Fabry Disease, 4D-310.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
